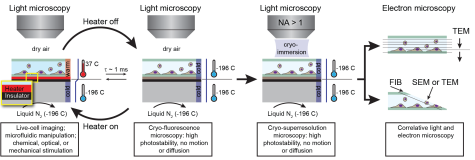
Despite rapid progress in the field, much of the potential of microscopy at cryogenic temperature today is still untapped due to limitations in sample preparation. First, vitrification technologies for cryomicroscopy have evolved only incrementally since the 1960s and cannot be combined with many of the sophisticated live imaging methods that have emerged over the past decade. Second, while the synergy of light and electron cryomicroscopy is extremely powerful, cryomicroscopy with light is still in its infancy. Finally, new technologies for ultra-rapid heating and cooling of single cells are needed to systematically advance our understanding of reversibility in the cryopreservation of e.g. stem cells, oocytes, or sperm cells.
This project aims to create a microfluidic technology platform for the direct vitrification of cells in the light microscope by ultra-rapid cooling with millisecond time resolution. The cells will then be imaged at high resolution using electron microscopy and advanced modes of light microscopy combined with new optics adapted to cryogenic conditions. Ultimately, we aim to elucidate if and under which conditions cryofixation can be reversed by ultra-rapid warming such that dynamic cellular processes resume unperturbed.
We expect that our research program will help to gain new insights into the structural and molecular basis of dynamic processes in cells. Understanding these connections is an important step toward identifying the causes of many diseases and devising effective therapies.
Integrated Micro-Nano-Systems (IMNS)

Microsystems for Cryomicroscopy
Microsystems for Cryomicroscopy
The ERC project MICROCRYO aims to vastly improve our ability to study dynamic processes in cells and organelles by developing new methods for correlating live-cell imaging, cryogenic light microscopy, and electron cryomicroscopy with millisecond temporal and nanometer spatial resolution.